AutoBuSTeD
Automated Bubble Sweat Test Diagnostics
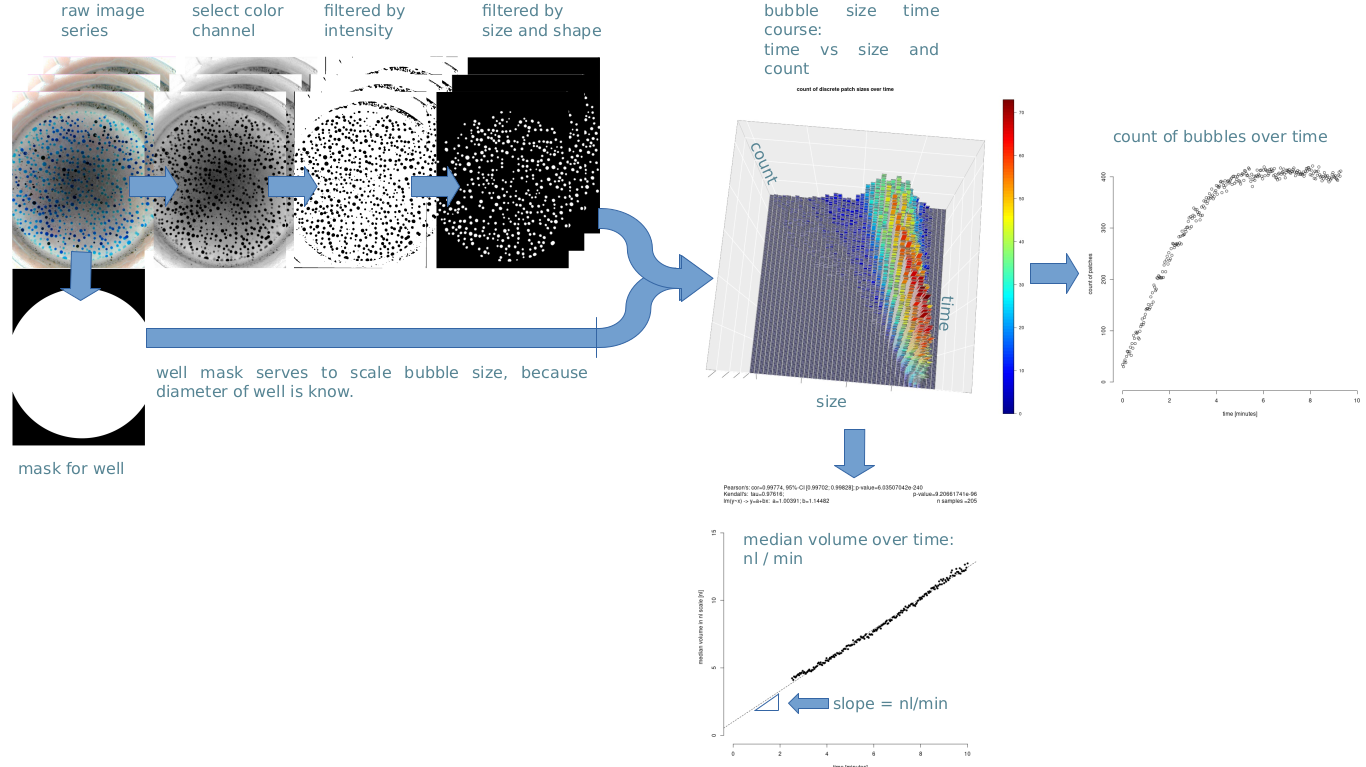
Aims of this collaborative project
This project aims to automate and standardize the analysis required for a diagnostic test for cystic fibrosis.
Our partner Teresinha Leal from the UCLouvain in Brussels has established and improved a non-invasive version of the imaging based sweattest with a linear characteristic identifying heterozygous as half responders useful to study genotype-phenotype correlation of rare CFTR mutations.
Another advantage of the new image-based test is that it has proven to be able to detect improved levels of CFTR function in patients treated with CFTR modulators. However, a severe practical limitation of the test is the manual image analysis (80 images/test), which is labour-intensive and can take 6 hours per test.
The Automated Bubble Sweat Test Diagnostics – AutoBuSTeD aims at overcoming the practical limitation of the new sweat test due to the labour cost of its data analysis.
Recently we successfully applied an enhanced imaging hardware setup developed at the UMG, which was intended to be used in conjunction with our AutoBuSTed software in a study held at the MHH in Hannover .
Web Resources
You can find additional information e.g. on the publically available software modules at our GitLab repository.
Funding
Our project was initially funded by the Christiane-Herzog-Stiftung via the Forschungsförderpreis für wissenschaftliche Nachwuchsförderung 2018 for our project proposal "Verbesserung der individuellen Bestimmung der CFTR-Restfunktion durch automatisierte Bildanalyse von beta-adrenerg-abhängiger Schweisssekretion"
In July 2022 we received the LIFT-OFF 2022 Gründungswettbewerb award of the Georg-August-University Göttingen - 2. place in the category science.
In October 2022 we received together with our partners from the Hannover Medical School (MHH) the decission notification that our next clinical study "The β-adrenergic sweat secretion test using AutoBuSTeD software as a novel diagnostic tool in patients with CFTR dysfunction" will be funded by the German Patient Organisations Mukoviszidose Institut gemeinnützige Gesellschaft für Forschung und Therapieentwicklung mbH small project funding program.
Project partners
The software is being developed transnationally and tested by our project partner with a cohort from Brussels (ClinicalTrials.gov Identifier NCT03584841) and from the Hannover Medical School in Hannover.
Department of Medical Bioinformatics (UMG) - Group of Chemoinformatics and Imaging
Dr. Manuel Nietert
Institut de Recherche Expérimentale et Clinique (IREC)
Louvain centre for Toxicology and Applied Pharmacology (LTAP)
Université catholique de Louvain (UCLouvain), Brussels, Belgium
Prof. Dr. Teresinha Leal
Department of Pediatric Pneumology, Allergology and Neonatology, Hannover Medical School (MHH), Hannover, Germany
Biomedical Research in Endstage and Obstructive Lung Disease Hannover (BREATH), German Center for Lung Research, Hannover Medical School, Hannover, Germany
Prof. Dr. Burkhard Tümmler,
PD. Dr. Anna Maria Dittrich,
Dr. Sophia T. Pallenberg
Publication
Pallenberg S. T., Held I., Dopfer C., Minso R., Nietert M.M., Hansen G., Tümmler B., and Dittrich A.M..
Differential Effects of ELX/TEZ/IVA on Organ-Specific CFTR Function in Two Patients with the Rare CFTR Splice Mutations c.273+1G>A and c.165-2A>G.
Frontiers in Pharmacology, Sec. Pharmacology of Ion Channels and Channelopathies, 14 (March 15, 2023). https://doi.org/10.3389/fphar.2023.1153656.
Pallenberg S., T.; Junge S.; Ringshausen C.; F., Sauer-Heilborn A.; Hansen G.; Dittrich A., M.; Tümmler B.; Nietert M.:
CFTR modulation with elexacaftor-tezacaftor-ivacaftor in people with cystic fibrosis assessed by the β-adrenergic sweat rate assay
Journal of Cystic Fibrosis,2021. https://doi.org/10.1016/j.jcf.2021.10.005
Press releases
Press release from the Mukoviszidose Institut – gemeinnützige Gesellschaft für Forschung und Therapieentwicklung mbH:
https://idw-online.de/de/news707760
Press release from the Georg-August-University Göttingen:
Patienteninformation - Suche nach Diagnostiktest
Für Patienten oder Angehörige, die auf der Suche nach einem Schweißtest zur Diagnose von Mukoviszidose sind und von einem Arzt oder Ärztin eine Überweisung erhalten haben, leiten wir Sie gerne an unsere klinischen KollegInnen der Mukoviszidose-Sprechstunde der UMG weiter. → https://herzzentrum.umg.eu/patienten-besucher/sprechstunden/ unter Mukoviszidose-Sprechstunde finden sie die aktuellen beiden Nummern.
Unser erweiterter Diagnostiktest aus diesem Forschungsprojekt wird bereits in der Region am Deutschen Lungenforschungszentrum an der Medizinischen Hochschule Hannover eingesetzt. Jedoch benötigen Sie für sich oder ihre Angehörigen eine Einschätzung mittels Standardtest, der die Chloridkonzentration bestimmt. Eine gute Erklärung zu diesem älteren Test finden Sie unter https://www.muko.info/mukoviszidose/diagnostik/schweisstest.
That might also interest you